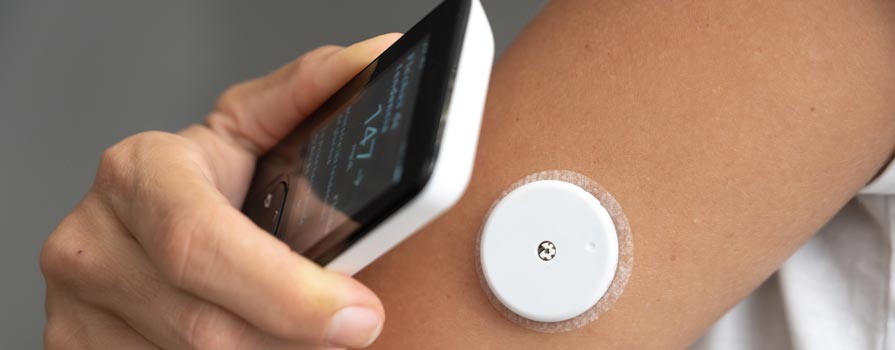
What are Continuous Glucose Monitoring Devices?
Continuous glucose monitoring (CGM) devices are small, wearable medical devices that track glucose levels throughout the day and night, providing ongoing data rather than single finger-stick measurements.
How do CGM Devices Work?
Sensor Insertion:
A small, flexible sensor is placed just under the skin and positioned in the interstitial fluid, not directly in a blood vessel.Glucose Detection:
The sensor uses an enzymatic reaction, commonly glucose oxidase, to detect glucose in the interstitial fluid. The reaction generates a tiny electrical signal proportional to the glucose concentration.Signal Conversion:
The device converts the electrical signal into a glucose value using built-in calibration algorithms.Wireless Transmission:
A transmitter sends glucose data wirelessly to a receiver or a compatible smart device.Continuous Updates:
he system records glucose readings every 1-5 minutes, creating real-time graphs and trend arrows, displaying glucose stability and directional changes.What are the Benefits of CGM?
- Real-time insights: CGM devices analyze daily patterns, trends, and the rate and direction of glucose changes, enabling users to make proactive decisions.
- Early warnings and alerts: continuous monitoring provides alerts for high or low glucose levels, including predictive alarms before levels become dangerous.
- Reduced finger-stick burden: CGM units reduce the need for frequent finger-stick tests, improving comfort and ease of use.
- Personalized feedback: continuous glucose monitors help users understand how specific foods, exercise, illnesses, or medications influence their glucose levels.
- Data sharing and remote monitoring: CGM devices enable secure data sharing with caregivers, guardians, and healthcare providers, supporting informed clinical decisions through detailed reports.
CGM Manufacturing Challenges and Boyd Solutions
Sensory Consistency and Accuracy:
CGM systems rely on enzyme-based electrochemical reactions to measure glucose, but sensor accuracy can drift with tiny variations in temperature, humidity, coating thickness, or material aging. Even micron-level inconsistencies can increase error rates and reduce sensor yield.
Boyd addresses this challenge through precision converting and tight process controls, that delivers foams, films, and adhesive components with consistent tolerances, supporting reliable and repeatable sensor performance.
Enzyme Stability and Shelf Life:
Glucose-sensing enzymes are biologically fragile and must remain stable during manufacturing, extended storage, and wear periods that last several days. Exposure to moisture, oxygen, or mechanical stress can degrade enzyme performance over time.
Boyd supports enzyme stability by selecting medical grade, ISO 10993-compliant materials and protective constructions that help minimize environmental exposure and preserve sensor performance throughout shelf life and use.
Biocompatibility and Skin Interaction:
Since CGMs stay attached to skin for long durations, manufacturers must manage irritation, allergic reactions, moisture buildup, and inflammation that can affect both comfort and sensor accuracy.
Boyd engineers biocompatible laminations using breathable materials and tuned adhesive architectures, that promote skin health while maintaining reliable device attachment.
Adhesion and Comfort:
CGM adhesives must withstand sweat, showering, and daily movement while still allowing for gentle, skin-friendly removal. Poor adhesion or discomfort can lead to premature device removal and reduced user adoption. Boyd balances adhesion and comfort by combining cushioning structures, barrier layers, and adhesive systems that maintain secure attachment without compromising skin integrity.
Miniaturization and Precision Assembly:
As CGM devices become smaller and more sophisticated, manufacturers face tighter tolerances, complex layer alignment, and higher precision requirements during assembly. These challenges increase the risk of defects and yield loss.
Boyd enables miniaturization through advanced clean room manufacturing, micro-precision converting, and controlled assembly processes that support high-volume production with improved yields.
Regulatory Burden and Slow Iteration:
Design or process changes in CGM manufacturing require re-validation, extensive documentation, and regulatory approval, which can slow innovation cycles and increase development risk.
Boyd reduces regulatory friction through regulatory-ready materials, robust documentation, and disciplined change-control processes that help manufacturers navigate FDA and global regulatory pathways more efficiently.
Why CGM Innovators Choose Boyd
With decades of medical manufacturing experience, deep materials expertise, and disciplined quality systems, Boyd helps CGM and medical wearable innovators bring reliable, patient-centric designs to market with confidence.
Contact Boyd today so our experts can recommend the effective processes and best fit materials for your continuous glucose monitor and medical wearable needs.